Acetaminophen For Back Pain
Acetaminophen Information Fda
Acetaminophen is a non-opiate, non-salicylate analgesic and antipyretic. the site and mechanism for the analgesic effect of acetaminophen has not been determined. the antipyretic effect of acetaminophen is accomplished through the inhibition of endogenous pyrogen action on the hypothalamic heat-regulating centers. Generic name acetaminophen drugbank accession number db00316 background. acetaminophen (paracetamol), also commonly known as tylenol, is the most commonly taken analgesic worldwide and is recommended as first-line therapy in pain conditions by the world health organization (who). 10 it is also used for its antipyretic effects, helping to reduce fever. 23 this drug was initially approved by the.
Acetaminophen is one of the most commonly taken medications in the united states, with an estimated 52 million consumers — 23% of adults in the country — using medications that contain this pain reliever each week, according to the consumer. Jun 28, 2016 · acetaminophen, also known as apap (in the united states), paracetamol (in europe and other areas of the world) or n-acetyl-p-aminophenol, is one of the most commonly utilized compounds worldwide; its use as an anti-pyretic or analgesic drug has been predominant since 1955, particularly due to the fact that it is easily accessible in various. Compare acetaminophen vs ibuprofen head-to-head with other drugs for uses, ratings, cost, side effects and interactions. acetaminophen is an effective mild pain reliever with a low risk of side effects. prescribed for sciatica, muscle pain,. Lists the various brand names available for medicines containing acetaminophen. find information on acetaminophen use, treatment, drug class and molecular formula.
Acetaminophen is the most commonly used painkiller in the u. s. but increasingly, researchers have questioned its safety. we look at the possible risks. whether in your handbag, a drawer at home, or your desk at work, chances are you have a. We may earn commission from links on this page, but we only recommend products we back. why trust us? acetaminophen is one of the safest drugs--provided acetaminophen structure you take it correctly. but in excess, it can seriously damage the liver. the issue is u.
Acetaminophen o-beta-d-glucosiduronic acid is a beta-d-glucosiduronic acid that is the o-glucuronide of paracetamol ( acetaminophen ). it has a role as a drug metabolite. it derives from a paracetamol and a beta-d-glucuronic acid. it is a conjugate acid of an acetaminophen obeta-d glucosiduronate. The ax form of acetaminophen is ax2, which means that the acetaminophen is linear. 180° 180° powered by create your own unique website with customizable templates.
Taking acetaminophen for fever or pain relief is generally safe and effective. but it can be harmful if not taken correctly. know the benefits and risks of acetaminophen and how to use it safely. you’ve probably taken acetaminophen at one t. Structure, properties, spectra, suppliers and links for: paracetamol, acetaminophen, 103-90-2. The basic amino group is written nh2–, though at times one or acetaminophen structure both of the hydrogen atoms are replaced by something else, as it is for tylenol. finally, “phen” stands for phenyl. remove one atom from a benzene ring and you have phenyl. c₆h₅is the simplest phenyl ring. Acetaminophen (tylenol) is a type of analgesic that decreases the brain&039;s perception of pain. it is commonly used for relief from back pain, mild arthritis pain, and headaches. acetaminophen (tylenol) belongs to a class of drugs called.
Acetaminophen is used by humans for pain and fever. is this drug safe for dogs? here's what you need to know before you give your dog acetaminophen. acetaminophen, or tylenol, is a common over-the-counter medication used by humans to reliev. Jul 02, 2020 · acetaminophen also selectively inhibits cox-3 enzymes. the antipyretic action of the drug is due to direct action of the drug on the heat-regulating centers in the brain. this results in sweating, vasodilation and loss of body heat. structure activity relationship. sar of acetaminophen can be summarized as follows:. Ibuprofen and acetaminophen are drugs for mild to moderate pain. learn how they compare. introduction acetaminophen and ibuprofen are medications used to treat pain and fever. however, they have some differences. acetaminophen belongs to a.
May 04, 2011 · a drug monograph is a detailed explanation of a drug. it contains information about the structure, function, uses, dosing, mechanism of action, side effects, adverse effects, etc of the drug. Figure 1: molecular structure of acetaminophen pk of acetaminophen is 9. 70 and is classified as a weak acid 3. acetamino phen is present in the for m of crystalline whi te powder, odorless, with a. Paracetamol, also known as acetaminophen, is a medication used to treat fever and mild to moderate pain. [11] [12] at a standard dose, paracetamol only slightly decreases body temperature; [11] [13] [14] it is inferior to ibuprofen in that respect, [15] and the benefits of its use for fever are unclear.
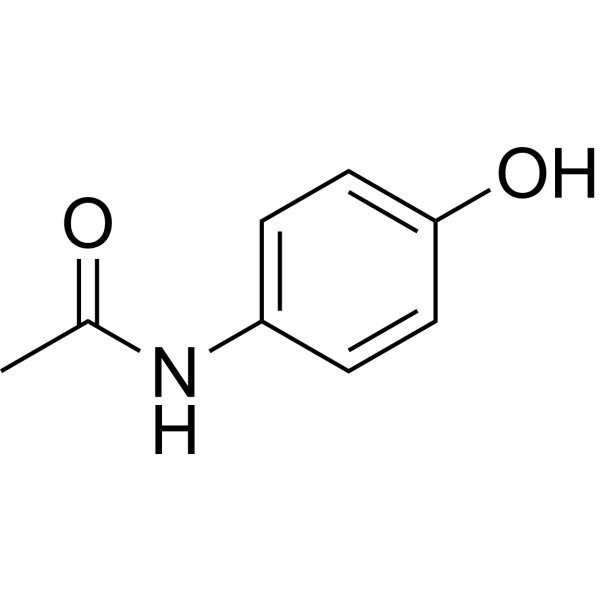
Acetaminophen is a p-aminophenol derivative with analgesic and antipyretic activities. although the exact mechanism through which acetaminophen exert its effects has yet to be fully determined, acetaminophen may inhibit the nitric oxide (no) pathway mediated by a variety of neurotransmitter receptors including n-methyl-d-aspartate and substance p, resulting in elevation of the pain threshold. Structure and reactivity paracetamol consists of a benzene ring core, substituted by one hydroxyl group and the nitrogen atom of an amide group in the para (1,4) pattern. the amide group is acetamide (ethanamide). Molecular formula. c8h9no2. synonyms. acetaminophen-d3. 60902-28-5. 2,2,2-trideuterio-n(4-hydroxyphenyl)acetamide. paracetamol;4'-hydroxyacetanilide;4-acetamidophenol;apap. paracetamol d3 (methyl d3) more. A chemical structure of a molecule includes the arrangement of atoms and the chemical bonds that hold the atoms together. the acetaminophen molecule contains a total of 20 bond (s) there are 11 non-h bond (s), 7 multiple bond (s), acetaminophen structure 1 rotatable bond (s), 1 double bond (s), 6 aromatic bond (s), 1 six-membered ring (s), 1 secondary amide (s) (aliphatic) and 1 aromatic hydroxyl (s).
Structure. find similar structures. molecular formula. c26h33n2o9p. synonyms. codrix. acetaminophen mixture with codeine phosphate. migraeflux-green. 67889-72-9. Acetaminophen: fda encourages the safe use of over-the-counter (otc) and prescription (rx) pain medicines. the fda has taken action to improve the safety of consumers when using acetaminophen. information for use during pregnancy, in childr. Acetaminophen may be the single most effective non-prescription medication for lower back pain and has the fewest side affects. acetaminophen (tylenol) is a medication that it is thought to relieve pain by working in the central nervous sys.
More acetaminophen structure images. Paracetamol, also known as acetaminophen, is a medication used to treat fever and mild to moderate pain. at a standard dose, paracetamol only slightly decreases body temperature; it is inferior to ibuprofen in that respect, and the benefits of its use for fever are unclear.
0 komentar:
Posting Komentar